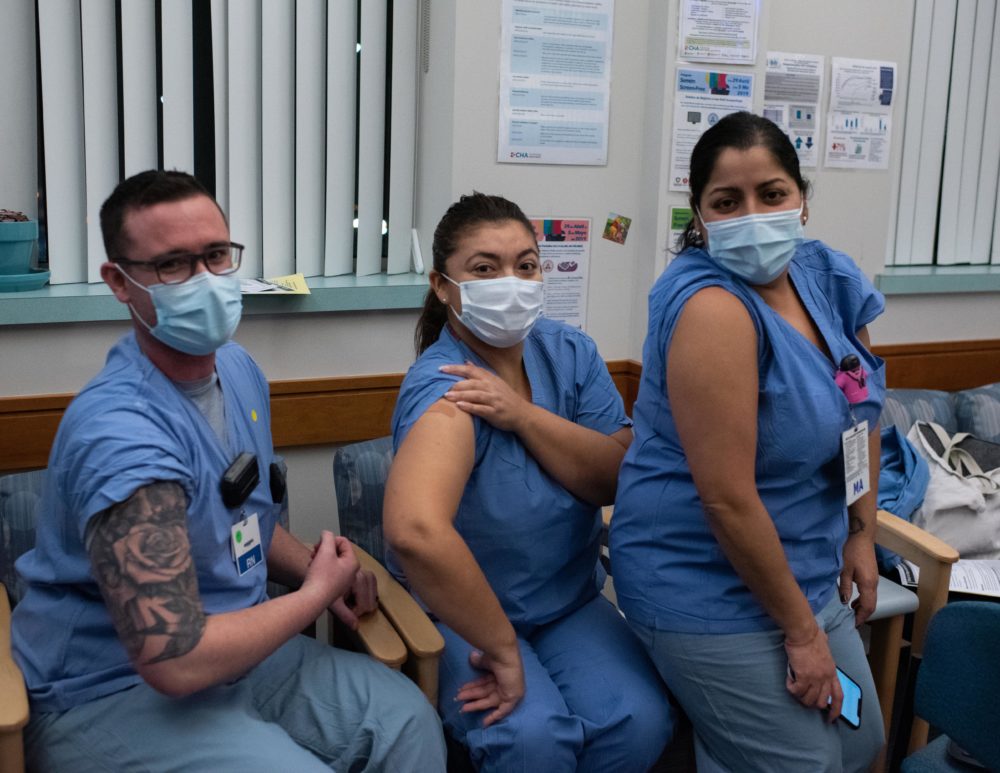
Last month, primary care doctor John Adams realized that coronavirus vaccines would be coming very soon, and he wasn’t ready for them. He didn’t know enough yet to counsel patients or make his own decision about whether to get one. And some of his colleagues at the Cambridge Health Alliance COVID-19 clinic in Somerville, Mass., felt the same.
“It was very much like, ‘We need to learn more about how these things work, because they’re coming fast’,” Dr. Adams says, “and we all sort of shared these doubts.”
They also shared unusually intense experience with COVID-19: The clinic has treated thousands upon thousands of patients since March, and its staff knows better than most how devastating the virus can be.
So Adams and physician assistant Neha Sandeep did some research. And at a meeting earlier this month, they presented their findings to the clinic’s medical staff — including their conclusion that although it took just one year instead of the usual 10 or 15 to develop the first coronavirus vaccines authorized for use, that didn’t mean corners had been cut.
“So these vaccines have been developed under a sort of a rapidly accelerated timeline,” Dr. Adams explained to colleagues. “So — if you go to the next slide — the way that that’s happened is basically by doing everything super fast and also sort of stacking these various phases.”
The presentation also addressed the newness of the mRNA technology the first two vaccines use: “Before going into this presentation, I was kind of worried about the idea [that with] these mRNA vaccines, you’re injecting genetic material into the cells,” he said. “I was worried: Could this cause cancer?”
“But actually safety is a big advantage of the mRNA vaccines,” Adams explained, because they trigger the immune system without the risk of using an actual virus to do it, and they have been tested for years in humans — though against cancer rather than viruses.
wbur.org/commonhealth/2020/12/30/somerville-clinic-coronavirus-vaccination
CHA physician Gerard Coste, MD provides a COVID-19 vaccine primer for healthcare and essential workers. This is intended for use by clinicians and leaders to share details about the Pfizer and Moderna vaccines, answer questions about their safety and support widespread adoption to help prevent the spread of COVID-19. Learn more via NPR affiliate WBUR:
wbur.org/commonhealth/2020/12/30/somerville-clinic-coronavirus-vaccination
Previously:
Subscribe to be notified of new posts:
![]()